Tracking nitrate and ammonium in the environment
Newly developed methodologies are improving our understanding of nitrogen cycling.
20/10/2022
At BGS’s Stable Isotope Facility, we are developing a suite of laboratory methodologies to analyse the key nitrogen-bearing species nitrate and ammonium. These methods will allow much lower concentration samples to analysed, helping improve our understanding of nitrogen cycling within the environment.
The nitrogen cycle
Alongside carbon and phosphorus, nitrogen is one of the key micronutrients critical to all life on Earth. It is a core component in amino acids, which, in turn, form the building blocks of all genetic material: DNA and RNA.
Nitrogen is the most abundant gas on Earth, making up 78 per cent of the air we breathe. Atmospheric nitrogen is converted by bacteria in the soil to ammonium (NH4+), in a process called nitrogen fixation. From here it is converted to nitrite (NO2–) and nitrate (NO3–), which can be taken up by plants, thus entering the food chain.
In addition to natural nitrogen fixation, humans apply nitrogen-rich fertilisers to soils to reduce natural nitrogen limitation and promote crop productivity.
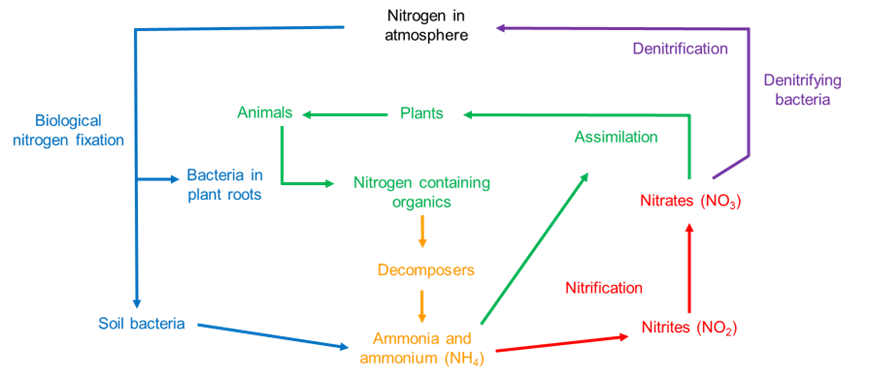
The nitrogen cycle, showing nitrogen fixation from nitrogen in the atmosphere to ammonium and the conversion of ammonium to nitrate before it is taken up into plants. Denitrification is the main process by which nitrogen is then lost back into the atmosphere. BGS © UKRI.
This application of fertilisers can have negative consequences. When nutrient-rich water runs off arable land and enters surface waters, for example lakes or rivers, it can promote uncontrollable algal growth. This can drastically decrease water quality, reducing the water’s oxygen content and leading to the death of aquatic species. Additionally, high concentrations of nitrogen can enter groundwater, from which it is almost impossible to remove, polluting aquifers that are traditionally used for water extraction and human consumption.
It is therefore critical that we understand how nitrogen enters our most precious environments, how it is cycled, transformed between different nitrogen species and released back into the atmosphere. To understand these processes, we need effective ‘tracers’. One of these is the stable isotope composition of nitrogen within different key molecules, including nitrate and ammonium.
New methods
The Stable Isotope Facility has carried out stable isotope analysis of nitrate and ammonium for many years. Traditional methods convert dissolved nitrate or ammonium to a solid, for example silver nitrate, allowing for the combustion of this solid within an elemental analyser coupled to a mass spectrometer. However, these methods required large volumes of sample water (more than 1 l) and high concentrations of either nitrate or ammonium. This used to limit the types and numbers of samples we could realistically analyse.
Recently, we have been working to improve in line with recent published methods. These new methods are up to a thousand times more sensitive than our old procedures, meaning we can analyse much smaller volumes of samples (1 to 4 ml) and at much lower concentrations (less than 1 mg-l). Unlike our old methods, these new techniques convert the dissolved nitrate or ammonium to nitrous oxide gas, which is then analysed by our trace gas and mass spectrometer system (Sercon Cryogas HS2022). This new mode of analysis has needed a lot of setting up and testing over the past year, but we are now getting great data for low concentration nitrogen species.

The Sercon and BGS team, who developed the Cryogas system currently used for N2O and other trace gas stable isotope analysis at BGS. BGS © UKRI.
The hope is that this faster, cheaper and more sensitive analysis will promote more wide-ranging nitrogen cycling studies using stable isotopes, helping to answer some of the key questions surrounding nitrogen pollution and sustainable fertilisation.
Acknowledgment
Thanks goes to the BGS Innovation Fund and Sercon for supporting this work.

Dr Andrew Smith
Isotope geochemist
Relative topics
Latest blogs

Strengthening capacity through partnership: a critical minerals perspective
28/04/2026
BGS has been working in partnership with the Geological Survey Department of Zambia (GSD) to build national capacity, improve data accessibility and support long-term, sustainable development.

PhD adventures in Copenhagen, Denmark: revealing past recovery processes of tropical forest systems through ancient environmental DNA
12/03/2026
PhD student Chris Bengt visited the University of Copenhagen to carry out very delicate extraction of aeDNA from lake-sediment cores, in the hopes of unlocking the secrets of past volcanic eruptions.

MARC Conference 2025: highlighting the importance of conferences to PhD students
16/02/2026
BGS and University of Nottingham PhD student Paulina Baranowska shares her experience presenting her research on nuclear forensics at her first international conference.

Hole-y c*@p! How bat excrement is sculpting Borneo’s hidden caves
23/12/2025
BGS researchers have delved into Borneo’s underworld to learn more about how guano deposited by bats and cave-dwelling birds is shaping the subsurface.

How the geology on our doorstep can help inform offshore infrastructure design
19/11/2025
BGS is part of a new collaboration using onshore field work to contextualise offshore data and update baseline geological models which can inform the sustainable use of marine resources.

Fieldwork on Volcán de Fuego
13/10/2025
Understanding how one of the world’s most active volcanoes builds up material, and how they collapse to feed hot flows

Esthwaite Water: applying novel approaches to understand lake-water nutrient pollution
19/09/2025
Andi Smith (BGS) and Savannah Worne (Loughborough University) embarked on fieldwork in the Lake District, applying a novel stable isotope method for tracing phosphorus sources.

Opening up the geosciences: making work experience more accessible
19/09/2025
BGS has been working with partners to make the geosciences more accessible to young people, including those from under-represented backgrounds.

PhD adventures in the Philippines: coring around Lake Bulusan
05/09/2025
Chris Bengt recounts his two-week field trip to Bulusan Volcano Natural Park in the Philippines to collect lake sediment cores, fresh soil and water samples.

Gemini: a new stable isotope tool
21/08/2025
BGS’s Stable Isotope Facility has new mass spectrometer equipment for analysing carbon and oxygen isotopes from carbonates and water.

BGS hosts India for ‘deep dive’ on carbon capture and storage
30/07/2025
Some of India’s top scientists visited BGS to explore the UK’s carbon dioxide storage research potential.

AI and Earth observation: BGS visits the European Space Agency
02/07/2025
The newest artificial intelligence for earth science: how ESA and NASA are using AI to understand our planet.





